Small Defects Have Large Impact
Max Planck materials scientists discover why ferroelectric materials sometimes lose their useful properties in the nanometer range
Many materials lose their useful properties as soon as their dimensions fall below a certain limit. This so-called size effect, the sources of which may be quite diverse, can be a road block for the miniaturization of electronic, electromechanic, and electrooptic components. For a particularly promising class of materials, viz. the ferroelectric oxides, researchers from the Max Planck Institute of Microstructure Physics have now identified a new origin of the size effect: Tiny linear defects, with an extension of less than about a tenth of nanometer, are able to deform a tube of material with rectangular cross section of about 4 by 8 nanometer around them. This deformation is so severe that the useful ferroelectric properties of the material are destroyed within the tube. This new finding shows that the formation of these defects has to be avoided, if ferroelectric oxides of nanometer dimensions are to be used as memory elements in future electronic components. (Nature Materials, Advanced Online Publication, January 18, 2004).
The ever increasing requirements on miniaturisation and efficiency of electronic components result in efforts to incorporate new materials into silicon-based microelectronics, in order to overcome the physical limits of classic materials. These new materials have to be employed from the outset with very small spatial dimensions. As a rule these dimensions are in the order of nanometers to tens of nanometers. One nanometer is the millionth part of a millimeter. In the nanometer range, the properties of most materials strongly depend on their outer dimensions, and the functionality of the material can even be lost below a certain size. Thus in basic materials science one tries to clearly identify the origins and mechanisms of this so-called size effect.
In case of a specific class of materials, the ferroelectric oxides, this challenge is particularly topical. Materials of this type find various applications in a remarkably broad spectrum of advanced electronic, electromechanical and electrooptic components. In many cases these materials tend to lose their functionality as soon as the outer dimensions fall into the nanometer range. Materials of this type are, e.g., lead zirconate titanate, strontium bismuth tantalate, and bismuth titanate. Ferroelectric oxides can be used to construct "non-volatile" memory cells in silicon-based microelectronics - cells that do not lose their information when the supply voltage is switched off.
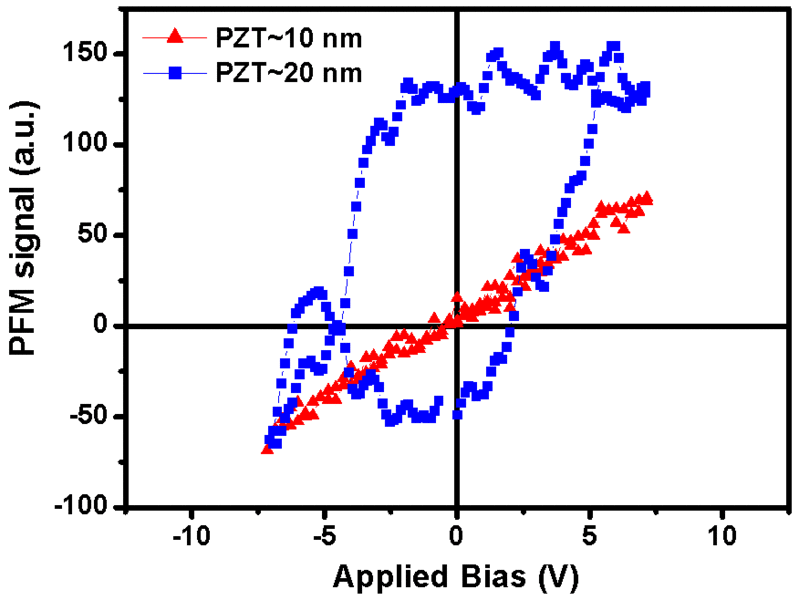
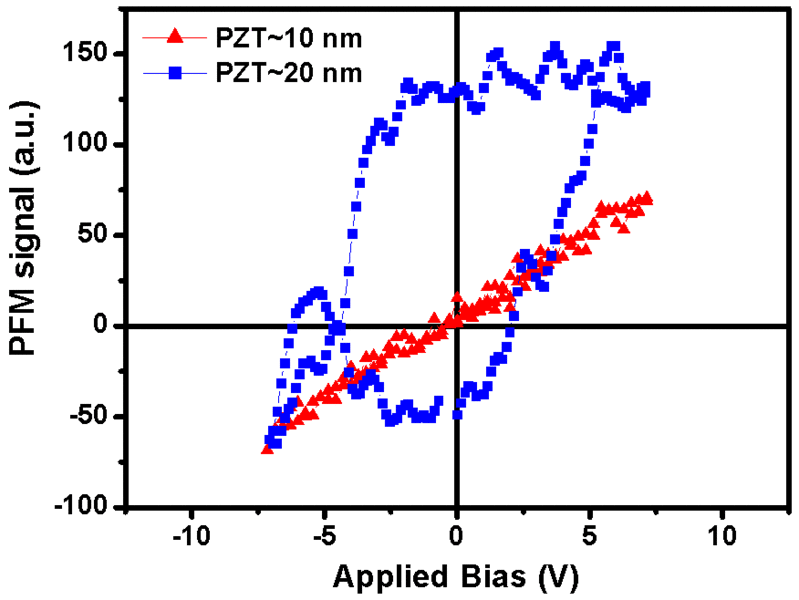
The working memories of personal computers -RAMs - could be significantly improved if it were possible to construct non-volatile memory cells with a storage density of several billion bits per square centimeter. In this way the time- and energy-consuming booting of the PC and the slow filesaving onto harddiscs could be avoided completely. However, in order to realize this goal a miniaturization of the memory cells down to a few dozens of nanometers is required. Consequently, the question why ferroelectric oxides lose their memory property below a certain size limit of nanometer range is particularly timely. Solid state physicists investigate this question worldwide, but a unified picture of the origin of size effects in ferroelectric oxides has not yet been attained.
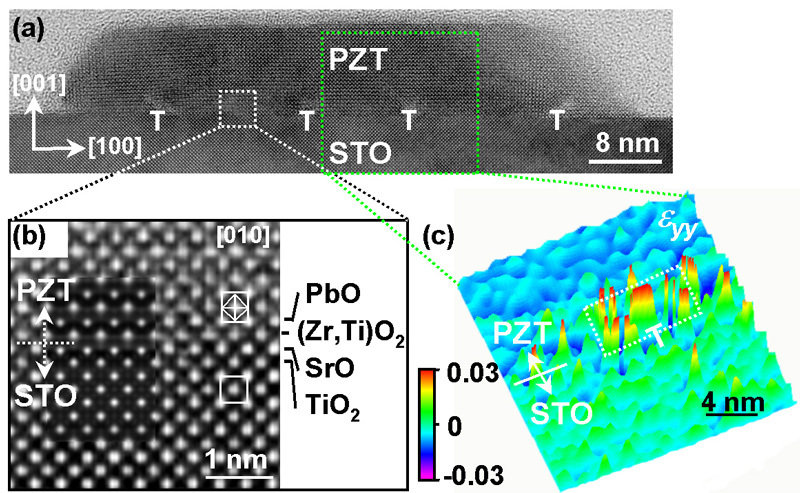
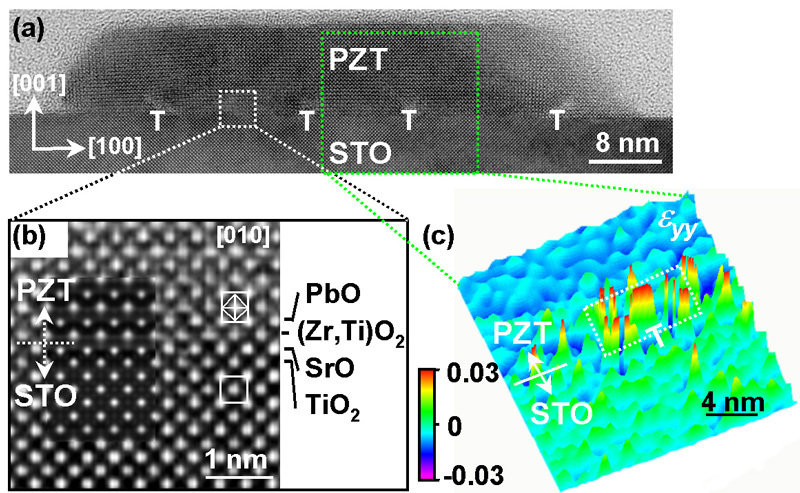
A research group around Ming-Wen Chu, Marin Alexe, and Dietrich Hesse at Max Planck Institute of Microstructure Physics in Halle (Saale) has now managed to identify a mechanism for the ferroelectric size effect which had not been considered so far. The Max Planck researchers were able to show that certain linear crystal lattice defects of less than a tenth of nanometer in diameter can siginificantly contribute to the loss of memory properties of 10 nanometer thin lead zirconate titanate islands. These defects form at the interface of the islands with the substrate made of strontium titanate. Applying quantitative high-resolution electron microscopy, the researchers were able to show that each of these linear defects, called misfit dislocations, is able to deform a tube of material with rectangular cross section of about 4 by 8 nanometer and with 20 to 50 nanometer length to such an extent that the material loses its storing property. If the ferroelectric island is so small that most of its volume consists of such tubes of deformed material, the island has no longer any storing properties. Thus the formation of such lattice defects has to be avoided, if ferroelectric oxides with nanometer-size dimensions are to be employed for storage properties.
"The perturbing impact of misfit dislocations on the optical and electronic properties of semiconductor nanostructures is a well known problem in semiconductor physics. Surprisingly, this problem has now turned out to be also most relevant to the memory property of ferroelectric nanostructures. This finding opens up new possibilities to systematically select well-defined combinations of materials for applications of ferroelectric oxides in miniaturized memory elements", says Professor Ulrich Gösele, director at Max Planck Institute of Microstructure Physics.